What Is the Heisenberg Uncertainty Principle? A Beginner's Guide to Quantum Uncertainty
Quantum mechanics explained simply for students, curious minds, and early researchers
The Question That Shook Physics
Imagine trying to take a photograph of a hummingbird in mid-flight. The faster you try to freeze the image, the sharper the shutter speed, the less information you capture about where it was heading. Now imagine that this isn’t just a photography problem. Imagine this is a law of the universe itself.
That is the spirit behind one of the most mind-bending ideas in all of science: the Heisenberg Uncertainty Principle.
It tells us that at the quantum scales, the world of atoms and electrons, nature has a built-in fuzziness. No matter how perfect your instruments are, no matter how advanced your technology becomes, there are pairs of properties that can not both be known precisely at the same time. This is not a measurement problem. It is a fundamental truth about reality in Quantum Mechanics.
In this guide, you’ll learn what the Heisenberg Uncertainty Principle really means, why it exists, what the math is telling us, and why it matters far beyond the physics classroom.
What is the Heisenberg Uncertainty Principle? for beginners
At its core, the Heisenberg Uncertainty Principle states that:
You can not simultaneously know the exact position and the exact momentum of a particle.
The more precisely you know where a particle is, the less precisely you can know how fast the particle is moving. And in which direction? And the reverse is equally true: nail down the momentum, and the position becomes a blur.
This was proposed by the German physicist Werner Heisenberg in 1927, and it became one of the cornerstones of quantum mechanics. But here’s what makes it so philosophically shocking: this uncertainty is not caused by clumsy instruments or human error. It’s not that we haven’t built good enough tools yet. The uncertainty is intrinsic to the particle itself. Nature simply does not allow both values to exist with perfect precision at the same time.
In classical physics, you could, in theory, know everything about a moving ball: where it is, where it’s going, how fast. In quantum physics, that idea collapses entirely. Electrons, photons, and other quantum particles don’t behave like tiny billiard balls. They exist in a cloud of probability, not a fixed location.
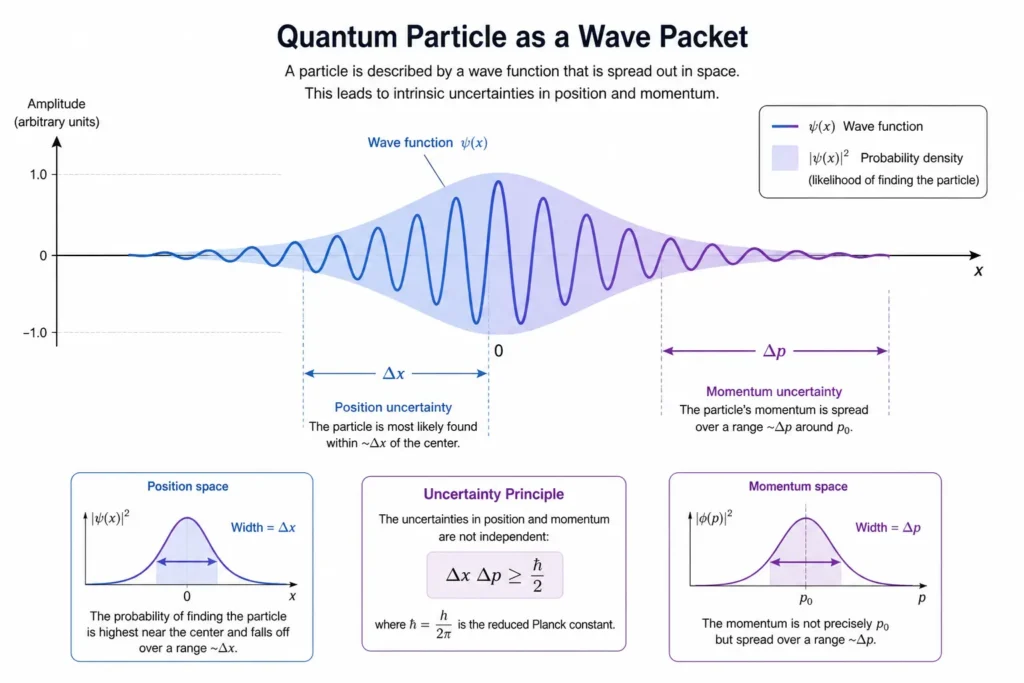
Explanation: This image shows a particle not as a hard dot but as a spread-out wave shape. The wider the wave is in space, the less certain the position; the narrower it is, the more uncertain the momentum. This visual captures the essential trade-off at the heart of quantum uncertainty — the more you squeeze one, the more the other spreads out.
The Mathematical Form of the Uncertainty Principle step by step for beginners
Now let’s look at the formula, don’t worry, we’ll walk through every symbol:
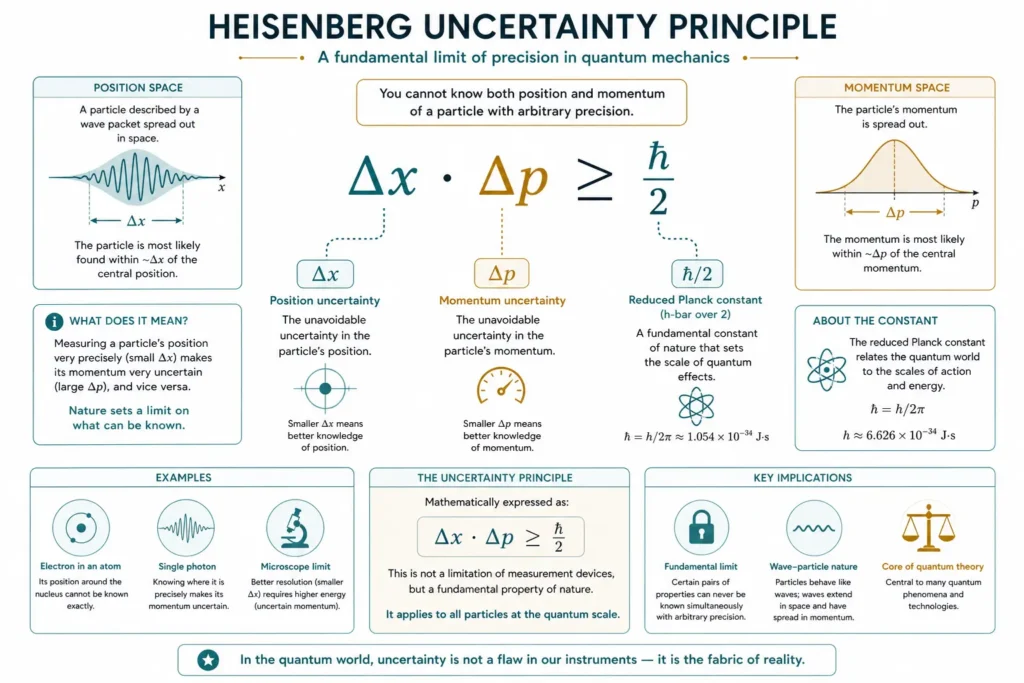
Δx · Δp ≥ ħ/2
Here's what each part means:
- Δx (Delta x) = the uncertainty in position: how spread out our knowledge of the particle’s location is
- Δp (Delta p) = the uncertainty in momentum: how spread out our knowledge of the particle’s motion is
- ħ (h-bar) = the reduced Planck’s constant, roughly 1.055 × 10⁻³⁴ joule in seconds, an incredibly tiny number that only matters at atomic scales
- ≥ = “greater than or equal to”: means the product of the two uncertainties can never be smaller than ħ/2
So what does the formula actually say? If you reduce Δx (you pin down the position more precisely), then Δp must increase to keep the product above ħ/2. You can’t shrink both sides at once. Nature enforces a minimum “budget” of uncertainty.
The reason ħ is so small is that we never notice this effect in daily life. For a basketball or a car, the uncertainty is there, but it’s so astronomically small compared to the object’s size that it’s completely irrelevant. It only becomes meaningful when we’re dealing with particles as tiny as electrons.
Why Does the Uncertainty Principle Exist? understand simply
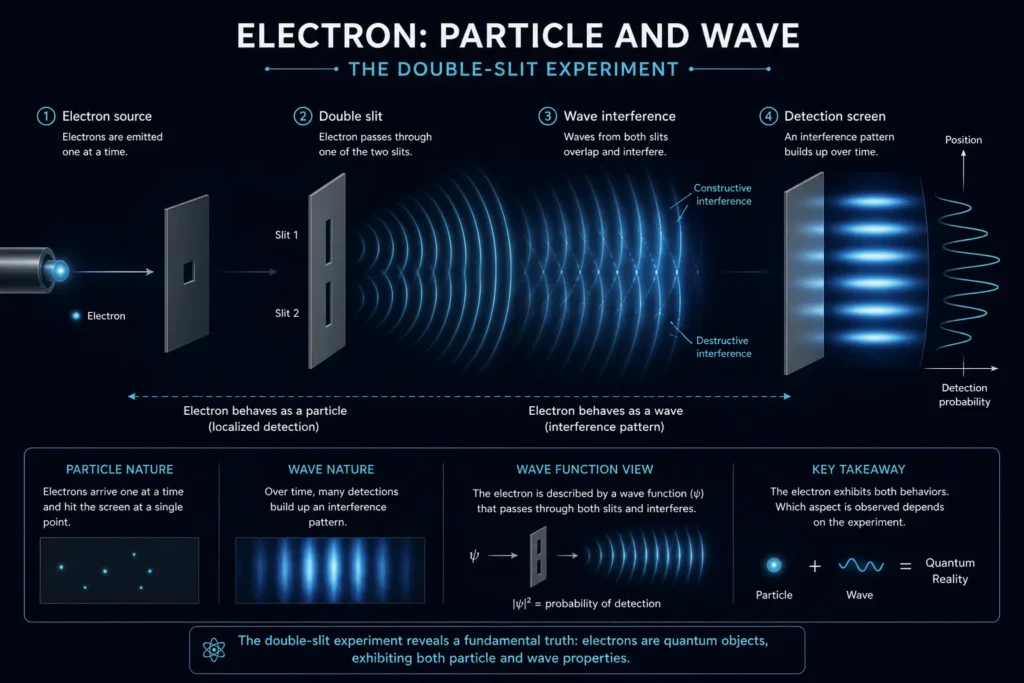
This is the big “why” concept, and the answer takes us into one of the strangest ideas in physics: wave-particle duality.
In the early 20th century, scientists made a startling discovery. The Light, which everyone knew was a wave, sometimes behaved like a particle. And electrons, which everyone assumed were particles, sometimes behaved like waves. French physicist Louis de Broglie proposed in 1924 that all matter has a wave nature. Every particle has a corresponding wavelength.
Here’s where the uncertainty comes from: waves, by their nature, are spread out. A pure wave with a perfectly defined wavelength (which corresponds to momentum) extends infinitely in all directions, and it has no fixed location. To give a wave a precise location, you have to combine many different wavelengths together, creating what’s called a wave packet. But once you do that, you’ve mixed together many momenta, so the momentum becomes uncertain.
It’s a mathematical fact about waves that there is no flaw in our instruments. This trade-off between knowing a wave’s position and knowing its frequency (momentum) is called the Heisenberg Uncertainty Relation in the context of quantum mechanics, but it actually arises from a universal mathematical property that applies to all waves.
So quantum uncertainty isn’t about observers disturbing particles (a common myth). It’s about the fact that particles are waves, and waves cannot have a perfectly defined location and a perfectly defined wavelength at the same time.
Classical vs. Quantum Uncertainty
To really appreciate how radical Heisenberg’s idea was, it helps to contrast it with the world of classical (Newtonian) physics.
In classical physics, the universe was like a perfectly wound clock. If you knew the position and velocity of every particle in existence, you could, at least in theory, predict the entire future of the universe. This is called determinism. Scientists believed that uncertainty was purely a practical problem: just build better instruments, and you could know everything.
In quantum physics, determinism falls apart completely. It’s not that our instruments are imperfect. The particles themselves don’t have a definite position and momentum at the same time. Before measurement, they exist in a superposition state, a haze of possibilities. The act of measurement doesn’t reveal a pre-existing fact; it forces the particle into one state from among many probable ones.
| Classical Physics | Quantum Physics | |
|---|---|---|
| Nature of particles | Definite position & momentum | Probabilistic, wave-like |
| Measurement problem | Practical limitation | Fundamental law of nature |
| Future predictability | Deterministic (in theory) | Inherently probabilistic |
| Uncertainty source | Imperfect tools | Intrinsic to reality |
This shifts from a clockwork universe to a probabilistic one is arguably the greatest intellectual revolution in the history of science.
Real-Life Examples of the Heisenberg Uncertainty Principle
Let’s bring this down to earth with some concrete examples of quantum uncertainty in action.
1. Electrons in Atoms
If electrons had perfectly defined positions and momenta (like classical particles), they would spiral into the nucleus almost instantly, releasing energy and causing atoms to collapse. The fact that they don’t know that atoms are stable is directly explained by the uncertainty principle. The electron cannot be confined to a point near the nucleus, because confining its position would give it enormous momentum, enough to escape. The uncertainty principle creates a minimum “spread” that keeps electrons in their orbitals and matter stable.
2. Quantum Tunnelling
Quantum tunnelling is the phenomenon where a particle passes through a barrier that it classically shouldn’t have enough energy to overcome. This happens partly because of position uncertainty, and a particle near a barrier has some probability of being found on the other side. This isn’t magic; it’s quantum mechanics at work. And it’s not just theoretical: nuclear fusion in the Sun depends on quantum tunnelling. Without it, stars wouldn’t shine.
3. The Stability of White Dwarf Stars
In stellar physics, the uncertainty principle contributes to the pressure that prevents certain dying stars from collapsing. As electrons are squeezed into a smaller volume (reduced Δx), their momentum uncertainty increases, generating a pressure called electron degeneracy pressure that holds the star up.
Why don't we notice it in everyday life?
Because the effect is proportional to ħ, Planck’s fantastically small. For a grain of sand, the uncertainty in position is smaller than the size of a proton by many orders of magnitude. It’s real, but completely imperceptible.
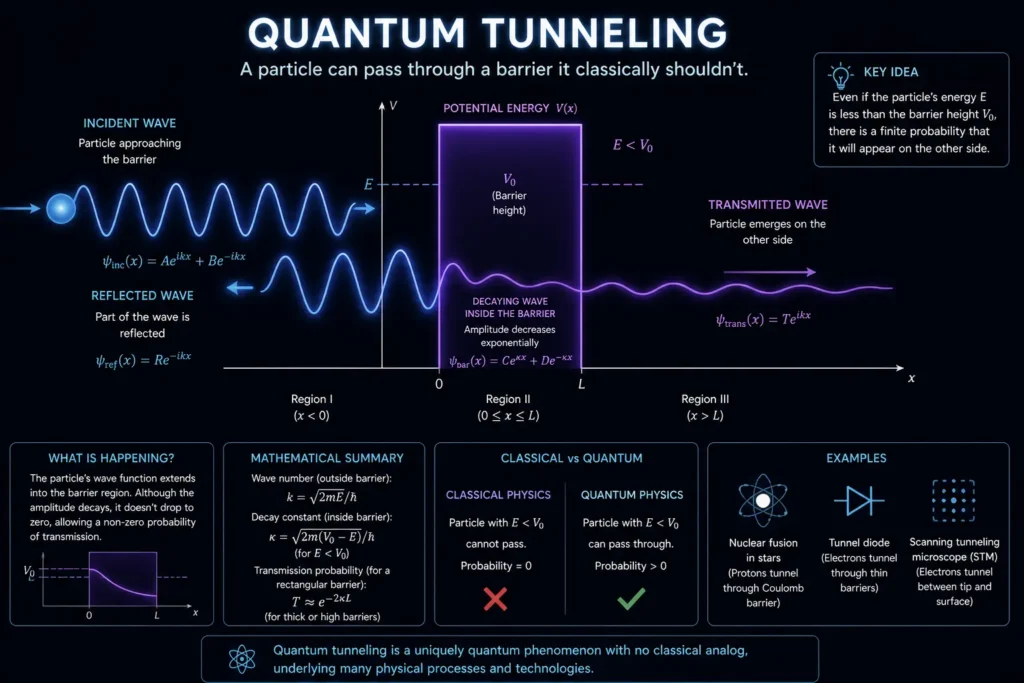
Explanation: This image shows a particle approaching an energy wall it shouldn’t be able to cross. Because its position isn’t precisely defined (thanks to quantum uncertainty), there’s a small but real probability of it appearing on the other side. This is quantum tunneling — and it powers the nuclear reactions inside the Sun.
Applications and Importance in Modern Physics
The Heisenberg Uncertainty Principle isn’t just an abstract philosophical idea. It has very real consequences for modern science and technology.
Atomic and Molecular Structure:
The entire framework of chemistry, why atoms are bonded, why molecules have specific shapes, is built on quantum mechanics, which rests on the uncertainty principle. Without it, we couldn’t explain why the periodic table looks the way it does.
Quantum Computing:
Quantum computers use the probabilistic, superposition-based nature of quantum mechanics to perform certain computations far faster than classical computers. The uncertainty principle is baked into the very behaviour of the qubits (quantum bits) these computers rely on.
Electron Microscopes:
Interestingly, the uncertainty principle also sets a lower limit on how precisely any quantum measurement can be made. This has practical implications for the design of electron microscopes and particle detectors.
Laser Technology and the Heisenberg Limit:
In precision measurement, like detecting gravitational waves with LIGO(Laser Interferometer Gravitational-Wave Observatory), scientists bump up against fundamental quantum noise floors set by the uncertainty principle. Understanding this limit has led to clever engineering solutions using “squeezed light” to push past it.
Fundamental Physics:
The uncertainty principle is what prevents quantum field theory (the framework that describes all known forces and particles) from producing infinite nonsensical results in many calculations. It’s woven into the fabric of how the universe works at the deepest level.
Common Misconceptions About the Uncertainty Principle
This principle is often misunderstood even by people who’ve heard of it. Let’s clear up the biggest myths.
❌ Myth 1: "It's just a measurement problem: we disturb the particle when we measure it."
This was actually Heisenberg’s original, somewhat imprecise explanation. Modern quantum mechanics tells us the uncertainty is deeper than that. Even in principle, even with zero disturbance, both values can not simultaneously exist. It’s not about the act of measurement; it is about the nature of the particle itself.
❌ Myth 2: "Better instruments would eliminate uncertainty."
No. No instrument, however perfect, can violate the uncertainty principle. It’s not a technological barrier, it’s a mathematical and physical law.
✅ Truth: It's a fundamental limit written into nature itself.
The universe genuinely does not allow both position and momentum to be perfectly defined simultaneously. This isn’t a gap in our knowledge, it’s a feature of reality.
An Intuitive Explanation for Beginners
Still feeling a bit lost? Here's the simplest way to think about it.
Imagine you’re trying to describe a wave in the ocean. If the wave is spread across kilometres of sea, you can say very precisely where it is, but it has no sharp starting point and ending point, so its exact “size” (related to wavelength) is hard to pin down. If instead you generate a very sharp, focused ripple in a pond, you can say where it is very precisely, but it contains a complex mix of frequencies.
That’s exactly the trade-off with quantum particles. A particle with very precise momentum is like a long, regular ocean wave spread everywhere. A particle with a very precise position is like a sharp ripple, a complex mix of many frequencies (momenta). Nature simply doesn’t let you have both at once.
Think of it like trying to clap your hands very fast while keeping them perfectly still. The two requirements are incompatible. Nature has built an incompatibility into the heart of reality itself.
Historical Background: Werner Heisenberg and the Birth of Quantum Mechanics
Werner Heisenberg was just 25 years old when he published his uncertainty paper in 1927 — a remarkable fact in itself. Working in Göttingen and Copenhagen alongside legends like Niels Bohr and Max Born, he was part of an extraordinary intellectual generation that dismantled centuries of classical physics in a matter of years.
Heisenberg developed his uncertainty relation while trying to understand how quantum measurements worked at a fundamental level. His insight that the mathematical structure of quantum mechanics inherently required complementary pairs of observables to trade off precision was revolutionary. It forced physics to abandon the classical ideal of a fully knowable, deterministic universe.
The uncertainty principle became one of the pillars of the Copenhagen Interpretation of quantum mechanics, which remains the most widely taught framework in physics. Heisenberg received the Nobel Prize in Physics in 1932 for “the creation of quantum mechanics.”
Summary: Key Takeaways
- What it is: The Heisenberg Uncertainty Principle states that the position and momentum of a quantum particle cannot both be known precisely at the same time.
- Why it exists: Because quantum particles are waves, and waves inherently cannot have a perfectly defined location and a perfectly defined wavelength simultaneously.
- The formula: Δx · Δp ≥ ħ/2: the product of position uncertainty and momentum uncertainty must always be at least ħ/2.
- It’s not a measurement problem: The uncertainty is intrinsic to nature, not a flaw in our tools.
- It matters: It explains atomic stability, enables quantum computing, drives nuclear fusion, and forms the foundation of modern physics.
- It only matters at tiny scales: For everyday objects, the effect is real but completely negligible
Frequently Asked Questions (FAQ)
What does Δx · Δp ≥ ħ/2 mean in simple terms?
It means that if you multiply the uncertainty in a particle’s position (Δx) by the uncertainty in its momentum (Δp), the result can never be smaller than a tiny fixed number (ħ/2 half of Planck’s reduced constant). In plain English: the more precisely you know where something is, the less precisely you can know how it’s moving, and vice versa.
Why can't we measure position and momentum at the same time?
Because quantum particles are fundamentally wave-like. A wave with a perfectly defined frequency (momentum) must extend infinitely in space it has no fixed position. A wave with a perfectly defined position must contain infinite frequencies, so its momentum is completely undefined. You can’t escape this mathematical reality.
Is the uncertainty principle true for all particles?
Yes, it applies universally to all quantum particles: electrons, protons, neutrons, photons, and even atoms and molecules. However, because Planck’s constant is so tiny, the effect is only significant for very small particles. For large, everyday objects, the uncertainty exists in theory but is unmeasurable.
Does the Heisenberg Uncertainty Principle affect everyday objects?
Technically: yes, but in practice, not at all. For a 1 kg object moving at 1 m/s, the minimum uncertainty in its position from the Heisenberg principle is roughly 10⁻³⁴ meters, many billions of times smaller than the diameter of a proton. You will never notice it.
Why is the uncertainty principle important in quantum physics?
It is one of the foundations of all of quantum mechanics. It explains why atoms are stable, why nuclear fusion can happen in stars, why quantum computers can exist, and why the subatomic world behaves so differently from the world we see around us. Without it, quantum physics as we know it simply couldn’t exist.
Conclusion: The Universe Is More Mysterious Than We Imagined
The Heisenberg Uncertainty Principle is not just a scientific formula, it’s a philosophical thunderbolt. It tells us that the universe, at its most fundamental level, is not a clockwork machine with fully knowable gears. It is a shimmering, probabilistic place where certainty has hard mathematical limits, and where the very act of asking “where is this particle?” and “how fast is it moving?” turns out to be a question nature refuses to answer in full.
What’s amazing is that this fuzziness isn’t a bug, it’s a feature. It’s why atoms hold together. It’s why the Sun burns. It’s why the rich, intricate chemistry of life is possible. The universe’s uncertainty, it turns out, is precisely what makes everything in it so wonderfully certain to exist.
Quantum mechanics is strange. Heisenberg’s principle is strange. But the universe was never obligated to be simple, and isn’t it more magnificent for not being so?


